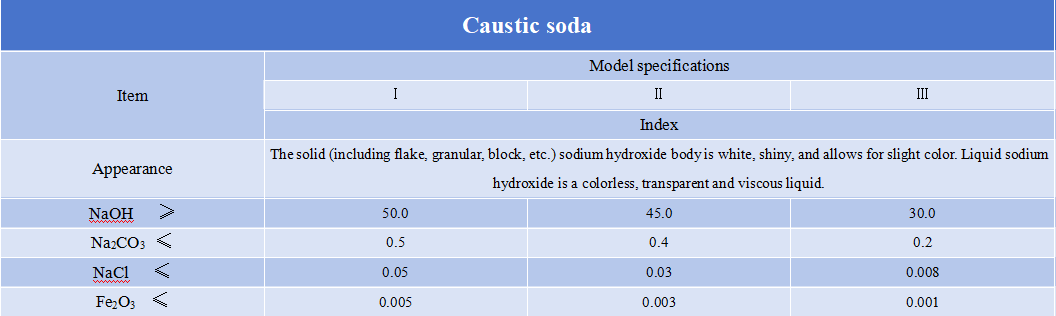
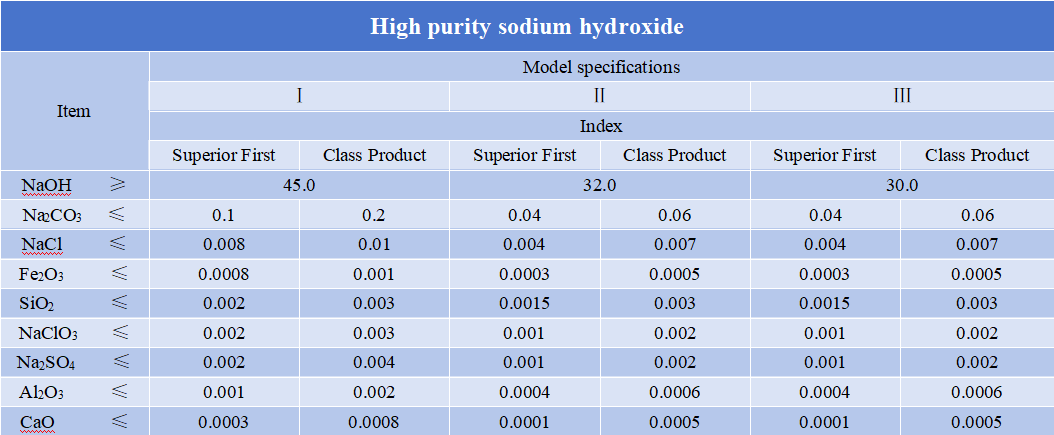
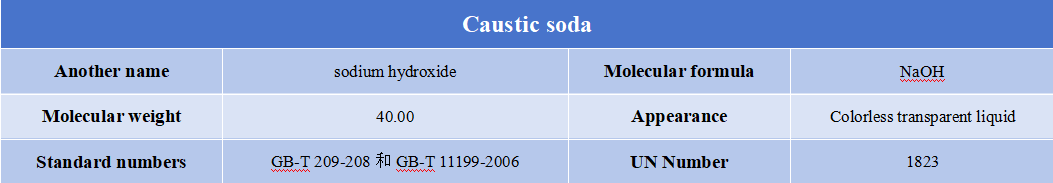
Caustic soda
Main purpose:
Sodium hydroxide is mainly used in the production of papermaking and cellulose pulp, as well as in the production of soap, synthetic detergents, synthetic fatty acids, and the refining of animal and vegetable fats. The textile printing and dyeing industry is used as a desizing agent, cooking agent, and mercerizing agent for cotton fabrics. The chemical industry is used to produce borax, sodium cyanide, formic acid, oxalic acid, phenol, etc. The petroleum industry is used to refine petroleum products and is used in oilfield drilling mud. It is also used for surface treatment in the production of alumina, metallic zinc, and metallic copper, as well as in glass, enamel, leather making, pharmaceuticals, dyes, and pesticides. Food grade products are used as acid neutralizers in the food industry, as peelers for citrus, peaches, etc., as detergents for empty bottles, cans, and other containers, as well as decolorizers and deodorizers.
When sodium hydroxide is used as a basic reagent, it can be used as a neutralizing agent, a masking agent, a precipitating agent, a masking agent for precipitation, an absorbent for small amounts of carbon dioxide and water, and a chromogenic agent for the determination of ketone sterols by thin-layer analysis. It is widely used in the manufacturing of various sodium salts, soaps, pulp, cotton fabrics, silk, viscose fibers, rubber products regeneration, metal cleaning, electroplating, bleaching, etc.
In cosmetics creams, sodium hydroxide and stearic acid act as emulsifiers through saponification, used in the manufacture of snow cream, shampoo, etc.